
اپنے رزق میں دوسروں کو شامل کریں
June 14, 202015 WhatsApp Features that are just amazing
November 10, 2020Coronavirus Vaccine update: What is actually going on? (Complete information)
In the current situation, where lockdowns and other preventive measures do not seem to stop the spread of the COVID-19, the coronavirus vaccine is the only solution for controlling the buildout of this pandemic. The vaccine itself is a preventive measure, but it works by preparing the immune system beforehand and making it identify the disease-causing virus, before the actual virus attacks. Human and medical history has proved that vaccination is an effective measure to control epidemics and diseases.
When the coronavirus vaccine is prepared, the spread of the disease can be controlled effectively. Experts state that if 60-70% of the population gets vaccinated, it is more likely that COVID-19 will not grow anymore, among the world population.
Coronavirus vaccine news: What are the scientists doing?
Coronavirus Vaccine has become the top priority of the scientists at this time and its development is going on at breakneck speed. About 198 research groups are working in different countries to accomplish this task, 44 of which are in clinical and 154 in the pre-clinical stage, as given by the corona vaccine by WHO updates. You can check out the names of the candidates and details at the WHO corona vaccine update section.
The development of a vaccine normally takes years, in order to get available to everyone. It has to pass through many phases and trials before declaring it safe for the use of the common population. The coronavirus vaccine, however, is a global emergency. Scientists are trying to complete this task as soon as possible, i.e. within 12-18 months after the virus was first identified.
To actually know what stage of development is the vaccine at, you need to get a little understanding of how the vaccine works.
corona vaccine details: How does the vaccine work?
A vaccine consists of a weakened or dead pathogen or parts (the immunogenic parts) of the pathogen. The vaccine interacts with the immune system of the body, faking a viral attack. The body considers it a real viral attack and an immune response is triggered, producing antibodies against the virus. Since it is a fake viral attack, the body does not develop a disease. On the bright side, it gets a kind of experience or memory to fight that particular virus. So, if the virus actually attacks, our immune system is ready and well-prepared to defend the body.
There is not one corona vaccine formula. Different research groups are taking different approaches to make the corona vaccine. Currently, there are 4 different methodologies being applied by scientists to make the vaccine.
- Weakened or dead virus vaccine: This type of vaccine uses the whole virus, the pathogenic part of which is either weakened or killed. It is the same method that was used to make the measles, mumps, and rubella (MMR) vaccine.
- Viral protein parts vaccine: This type of vaccine only uses the immunogenic part of the virus i.e. the viral surface proteins. Our immune system responds to it in the same way as it does with the whole virus.
- Virus Vector vaccine: This vaccine type uses adenovirus (a virus that infects monkeys and not humans) as a vector or transporter of those genes of coronavirus that decode to form the viral proteins. Once in the blood, our body itself translates the genes into viral proteins, which in turn, trigger an immune response.
- Viral mRNA vaccine: This kind of vaccine works by injecting viral mRNA into the body. The mRNA is directly translated into viral proteins, to interact with the immune system.
Corona Vaccine status: Corona Vaccine human trial
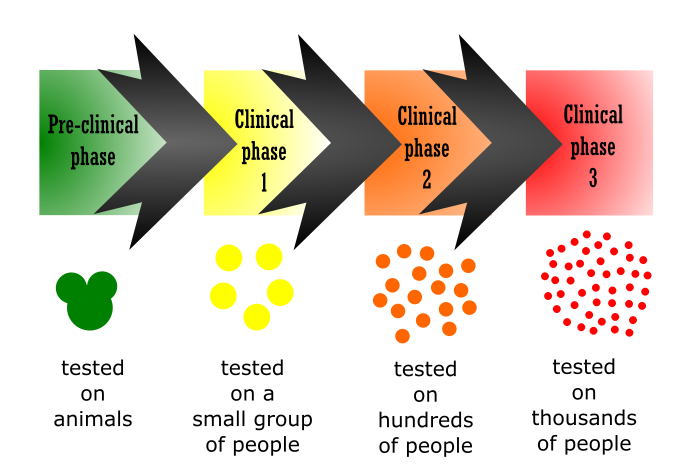
Much care and precaution are taken before a vaccine is finally launched. After production, it goes through 4 phases of testing to ensure that it is safe for the human body.
The pre-clinical phase: The vaccine is given to animals to check if it stimulates the immune system or not.
Clinical phase 1: The vaccine is administered to a small group of people, to know if it is safe and to study its effects on the human body.
Clinical phase 2: If the phase 1 trial is successful, then the vaccine is given to hundreds of people. The safety and dosage of the vaccine are determined at this phase.
Clinical phase 3: The vaccine is finally given to thousands of people in the population. This is a multi-cultural check and a step for further ensuring the safety of the vaccine. If it is successful, the vaccine is approved for public use.
Corona vaccine trials are going on around the globe, many of which are in phase 3 of clinical testing. There is even news about ‘corona vaccine approved’ in Russia), which is without the completion of phase 3.
Corona vaccine company: Who is making the vaccine?
Many institutes and companies are working on the production of the coronavirus vaccine. The leading ones that are in phase 3 of clinical trials are CanSino Biologics, AstraZeneca, IQVIA, The university of oxford, Sinovac, Serum Institute of India, Johnson & Johnson, Moderna, Wuhan institute of biological products, Sinopharm and Novavax.
Corona vaccine expected release:
The corona vaccine expected release is by the end of 2020 to the mid-2021. People are hopeful that the corona vaccine date launch will be announced soon, once it passes phase 3 of the clinical trial. To date, there have been 1620 trials of the Corona vaccine.
Even after passing the phase 3 trial, there are other hurdles that need to be overcome. There needs to be a method for such a large-scale production and storage of the vaccine, the approval of regulatory authorities is required and many other small things that need to be considered. It is after this that the coronavirus vaccine is available in the market.
Corona vaccine update Pakistan:
In Pakistan, no known research is going on for the development of the vaccine. However, Pakistan is participating in phase 3 trial of Chinese Coronavirus vaccine Adenovirus Type 5 vector (Ad5-nCoV) made by CanSino Biologics and Beijing Institute of Biotechnology China (BIB), after the approval of the Drug Regulatory Authority of Pakistan (DRAP), on the recommendation of the NIH.
Corona vaccine trial in Pakistan:
The Adenovirus Type 5 vector (Ad5-nCoV) is a vaccine that is in its 3rd phase of trials. It is made by the CanSino Biologics and Beijing Institute of Biotechnology in China. Pakistan is enrolling thousands of its citizens for this trial. It started with 200 volunteers from Karachi at Indus Hospital Karachi. Now, the corona vaccine trial in Pakistan has been extended to other parts of the country in different private and government sector institutes like Agha Khan University Hospital, Shaukat Khanum Memorial Cancer Center Hospital & Research Center, University of Health Sciences, and Shifa International Hospital.
This participation will prove fruitful for the country. It has enabled Pakistan to participate in the global effort of vaccine production. Also, if this trial is successful, the vaccine can be delivered to its citizens at an economical price.


